 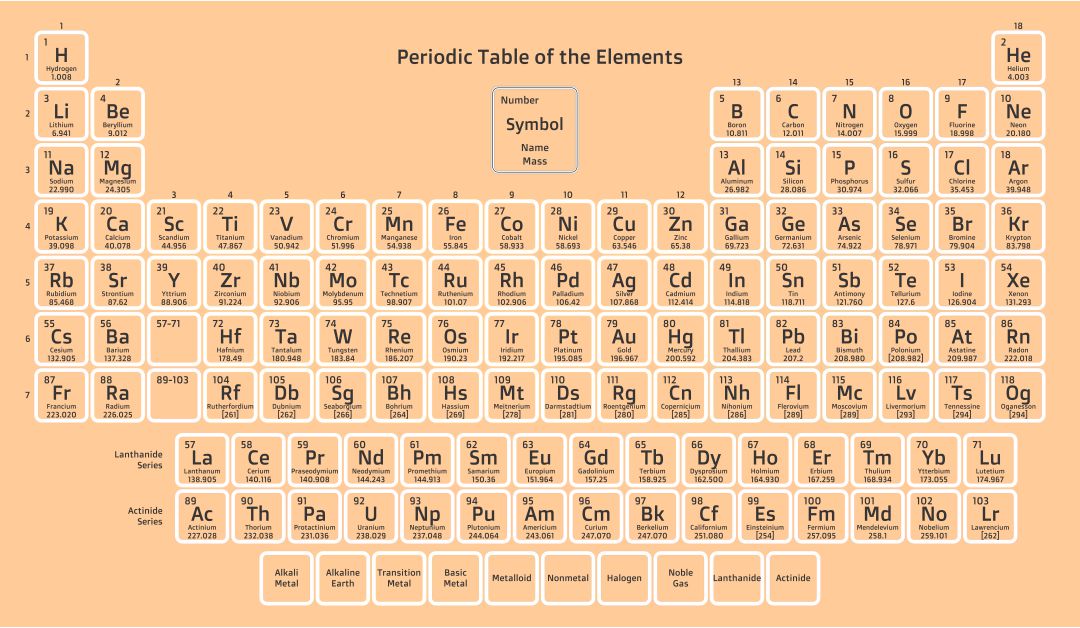
By losing this electron the lithium atom becomes a lithium ion, which is represented as Li +. For example, lithium has one electron in its outer shell which it needs to lose. We will need two hydroxide ions to exactly cancel the 2+ charge on Ca 2 +. Metal elements in groups 1, 2 and 3 of the periodic table all lose electrons to become metal cations. From our table, we know that hydroxide has the formula OH and that it carries a 1- charge. Use a periodic table to determine the charges achieved upon ionization of main group elements. This is to show that the subscript applies to the entire polyatomic ion. Calcium is an alkaline earth metalGroup 2 on the periodic tableso it forms ions with a 2+ charge. For example, iron(II) has a 2+ charge iron(III) a 3+ charge. acetate PERIODIC TABLE OF IONS arsenate arsenite benzoate borate bromate carbonate chlorate chlorite chromate cyanate cyanide dichromate CH3COO AsO4 3 AsO3 3 C6H5COO BO3 3 BrO3 CO3 2 ClO3 ClO2 CrO4 2 CNO CN Cr2O7 2 oxalate perchlorate periodate permanganate peroxide phosphate pyrophosphate sulfate. Group 2 alkaline earth metals lose two electrons and have a. Group 1 alkali metals all lose one electron and have a positive one charge when they become an ion. The metals, on the left side of the periodic table, will lose electrons. 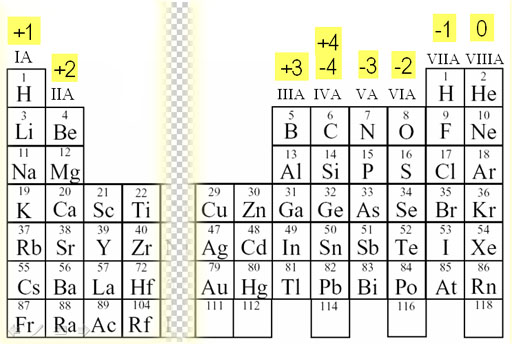
If more than one of a particular polyatomic ion is needed to balance the charge, the entire formula for the polyatomic ion must be enclosed in parentheses, and the numerical subscript is placed outside the parentheses. 3 shows how the charge on many ions can be predicted by the location of an element on the periodic table. Roman numeral notation indicates charge of ion when element commonly forms more than one ion. Many of the charges for ions formed by the main group elements can be predicted using the periodic table. The rule for constructing formulas for ionic compounds containing polyatomic ions is the same as for formulas containing monatomic (single-atom) ions: the positive and negative charges must balance. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
